Finish Your Trial Faster
Use Viomedo's end-to-end solution for patient recruitment and benefit from our unique strengths in medicine, technology and marketing.
Increased number of randomized patients per site
Total enrollment milestone reached faster
Success-based pricing per patient
Deep understanding of your protocol
Just like every patient, every clinical trial is unique. For a recruitment campaign things, such as the run-in phase, distinct cohorts and standard of care needs to be considered.

Before we start with any recruitment campaign, we make sure to understand your trial protocol and sites. The distinct requirements are then passed on to every module of our solution, in order to make sure that we can reach as many patients as possible, refer only the eligible ones to the sites and make sure that every patient is handled effectively and compassionately.
Experience in your indication area
You can rely on our experience across a variety of
indications
and therapeutic
areas.
CNS
Parkinson's, MS, Alzheimer's, etc
Endocrinology
T2DM, DKD, DME, etc
Inflammation
Crohn's, Colitis, Psoriasis, etc
Oncology
Colorectal, Lung, Prostate, etc
Respiratory
Asthma, COPD, IPF, etc
Women's Health
Endometriosis, PCOS, etc
Let us tell you more
Leverage our digital scale and experienced team to keep your trial on time and on budget.
Contact Uspre-screened
patients in H2 2017
clinical trials platform
in
Germany
and Austria
of combined experience
in medical
research
Making your trial broadly accessible
Sure, your trial is listed in the official registers,
but patients, caregivers
and potentially referring physicians do not use them.

We think patient-first and develop a clear, easy-to-understand study page for your trial based on the information architecture we have developed together with patients and patient advocacy groups across several indications. You will receive the complete dossier for the EC / IRB containing all recruitment material within just 8 days.
Having submitted material to most ECs in Germany (and several in Austria), we’re very confident that the material will be approved within the 1st submission.
Viomedo's customers include the top pharmas, biotechs and CROs



Reaching patients everywhere
Running ads in buses or Social Media is not adequate to start a meaningful interaction with patients. This is why our patient engagement is tailored to their needs.
Our patient recruitment approach is based on our unique distribution network that brings together the leading clinical trials platform, patient communities, health portals and advocacy groups. Targeted digital marketing techniques are used to supplement and broaden the outreach.
Over 95% of randomized patients were not
treated at the site
before.
Proprietary distribution meets precision marketing

Viomedo Platform
Our patient-facing online platform connects patients with over 2.000 clinical trials. It is the destination number 1 for patients looking for current research.
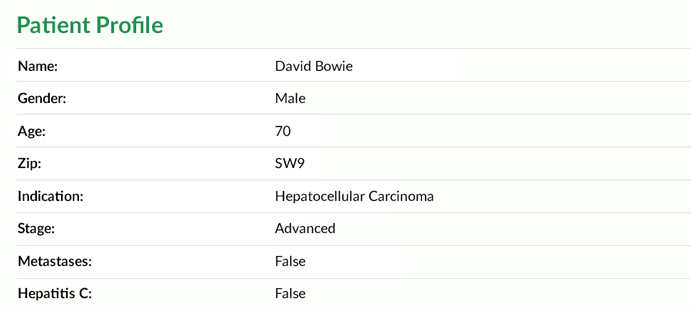
Viomedo Patient Database
More than 25.000 patients have registered with us in order to learn about clinical trials opportunities. More and more patients join every day.

Patient Advocacy Groups
We work together with an increasing number of patient advocacy groups across several indications.
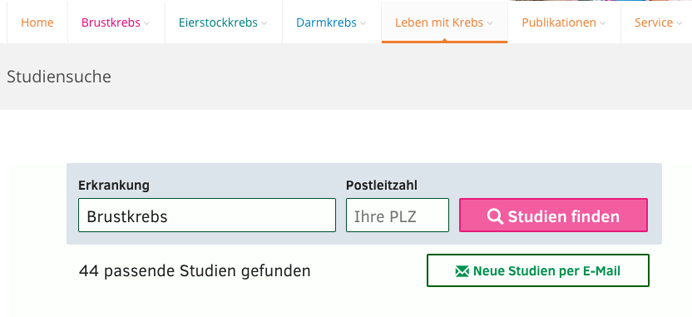
Health Portals
Viomedo is directly integrated on leading health portals, reaching patients wherever they seek health information.

Targeted Marketing
Leverage our vast marketing experience across a variety of channels reaching patients on all devices, nationwide.

As seen in
Patient-Trial Matching -
A patient-centric approach
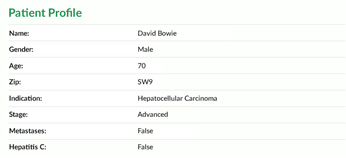
Clinical trials have an increasing number of inclusion and exclusion criteria. Thus, matching the right patient to the right trial is key for both patients and investigators.

Viomedo uses IRB approved digital pre-screeners that are tailored to the study indication and patient population. Our system has matched over 30.000 patients in the first half of 2017 with the right trial for them.
The pre-screener is just the first step, though. After a patient has passed the pre-screener, our medical team contacts them personally.
We call them in order to make sure that their information is accurate, ask additional questions, confirm their interest in the trial as well as motivation and explain the process. Only qualified patients are then referred to a site.
Site Support - Closing the gap
Even within one trial, there are significant differences between sites when it comes to process quality, performance and knowledge of the protocol. This is why we provide sites support tailored to their needs.

Our site team is led by study nurses and coordinators who speak the site’s language. By working closely with the investigator and her team, we support each site individually with the process from receiving to enrolling the patient.
Because we track the whole recruitment process for every patient, we can identify and address problems quickly and help sites convert referrals into randomized patients.
Recognized Innovation Leader

Success-based pricing per patient
Whether Big Pharma, emerging biotech or CRO, you only pay for actual patients enrolled. We have the processes, experience and SOPs in place to contract with all types of organizations and business units, whether local or global.

Our recruitment process is as transparent as our pricing. We provide all stakeholders with visibility into the overall flow of patients in real time through our portal.